Tetrabenazine – A forgotten drug for Dystonia
An 8 year old girl presented to the pediatric ER with complaints of fever for 1 week, altered sensorium for 5 days and multiple episodes of generalized tonic clonic seizures for 4 days. She was a known case of neurocysticercosis for the last 6 years, had been under antiepileptic therapy (carbamazepine and phenytoin) for 3 years and was discontinued after a seizure free period of 2 years. She was admitted to the intensive care unit for control of refractory seizures and was started on phenytoin, phenobarbitone, sodium valproate and a continuous infusion of midazolam after which her seizure episodes were abolished.
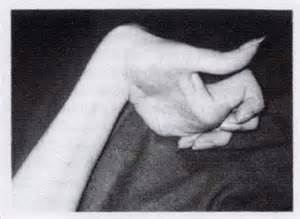
She developed a new onset of abnormal episodic body movements which comprised of sustained stiffness of neck, upper and lower limbs without any desaturation or tachycardia. Clinically these movements resembled dystonia.
Her CSF was normal, MRI head showed acute encephalitic changes. She was started on trihexyphenydyl and doses were gradually increased upto 30 mg which did not prove effective. Diazepam would contol her acute episodes but did not decrease the frequency of dystonias.
So, a trial of tetrabenazine was adopted and a dose of 12.5 mg was started. Her clinical condition dramatically improved with amelioration of dystonias.
Discussion:
Dystonia is characterized by involuntary sustained muscle contractions resulting in abnormal postures.Dystonia can affect any part of the body or specific groups of muscles, with the most common areas affected being the cranial-cervical region. It may also affect the extremities, trunk, facial, and vocal muscles.
Dystonia is often classified by anatomy (general, hemi-, segmental, multifocal, or focal). Generalized refers to widespread dystonia involving the legs and other areas. Hemi-dystonia affects the arm and leg on the same side. Segmental refers to dystonia affecting >2 adjacent focal areas but sparing the legs. Multifocal refers to dystonia affecting_2 separate focal areas, and focal refers to dystonia affecting only 1 specific focal area. Blepharospasm, cervical dystonia, oromandibular dystonia, spasmodic dystonia, and writer’s cramp are examples of focal dystonias.
TBZ (chemical name: 1,3,4,6,7,11b-hexahydro-9,10-dimethoxy- 3-(2-methylpropyl)-2H-benzo(a)quinolizin-2-one)(1). Commercially available TBZ is a racemic mixture of the RR and SS isomers. After oral administration,TBZ is rapidly absorbed within 1 hour; however, the systemic bioavailability of the parentdrug is low (5%) due to a high first-pass metabolism. (2 )
The primary pharmacologic action of TBZ and its active metabolites is to inhibit the human vesicular monoamine transporter 2 (VMAT2). The function of VMAT is to transport monoamines (eg. dopamine, serotonin, norepinephrine) from the cellular cytoplasm into synaptic vesicles. There are 2 human isoforms of this transporter: VMAT1 and VMAT2. VMAT1 is primarily in peripheral endocrine and paracrine cells. The VMAT2 transporter is predominantly expressed in the brain.21 Inhibition of VMAT2 prevents translocation of neurotransmitter monoamines from the cytoplasm into synaptic vesicles; therefore, the monoamines are exposed to premature degradation.(3) In addition to VMAT2 transporter inhibition, TBZ also alters serotonin 5-HT2 and beta-adrenoceptor density and is a weak postsynaptic dopamine-2 receptor antagonist.(4)
In a retrospective study, Jankovic and Beach reported results of TBZ therapy in 400 patients (pediatric and adult) with severe hyperkinetic movement disorders. Within this cohort, 190 patients had various dystonias (idiopathic cranial, cervical, and generalized dystonic disorders; tardive dystonias). Clinical response to TBZ was rated on a global response scale (GRS) from 1 to 5 (1 – marked reduction in abnormal movements and excellent improvement in function; 2 – moderate reduction in abnormal movements, very good improvement in function; 3 -moderate improvement in abnormal movements, only mild or no improvement in function; 4 – poor or no response in abnormal movements or function; and 5 – worsening of movement disorders, deterioration in function, or both). Patients with idiopathic dystonia (n =108) received TBZ for a mean (SD) of 29 (31.87) months and reached a maximum dose of 112.15 (58.94) mg/d.
Patients with tardive dystonia (n =82) received TBZ for a mean of 32.19 (39.28) months and reached a maximum dose of 125.15 (72.85) mg/d. Of the patients with idiopathic dystonia (47 with cranial distribution, 41 generalized, and 20 cervical), there was no difference in TBZ effectiveness based on affected area of dystonia (P values not reported).Patients with tardive dystonia showed more of an improvement than those with idiopathic dystonia. In patients with idiopathic dystonia, 62.9% had an initial response score of 1, and this finding fell to 45.3% at the final visit. In patients with tardive dystonia, 80.4% had a response score of 1 at the initial visit, with 73.1% having a score of 1 at the final visit.(5)
In another retrospective analysis, Kenney et al assessed the long-term efficacy of TBZ in 448 patients with various hyperkinetic movement disorders. Of this group, 132 patients (pediatric and adult) had dystonia. Mean (SD) age at initial TBZ treatment was 53.1 (1.7) years (range, 5.6–87.6 years), and mean duration of
TBZ treatment was 3 (0.4) years (range, 0.3–21.6 years). Consistent with other studies,68% of patients with dystonia experienced a marked or moderate improvement (assessed by using the GRS). TBZ has been used in pediatric and adult patients with various dystonias. (6)
Overall, based on the available prospective and retrospective data, TBZ demonstrates efficacy for dystonia but should be considered as an alternate treatment option to established therapies (eg, anticholinergics, botulinum toxin, muscle relaxants).(6)
References: 1. Jankovic J, Clarence-Smith K. Tetrabenazine for the treatment of chorea and other hyperkinetic movement disorders. Expert Rev Neurother. 2011;11:1509–1523 2. Roberts MS, McLean S, Millingen KS, Galloway HM. The pharmacokinetics of tetrabenazine and its hydroxy metabolite in patients treated for involuntary movement disorders. Eur J Clin Pharmacol. 1986;29:703–708. 3. Kanner BI, Schuldiner S. Mechanism of storage and transport of neurotransmitters. CRC Crit Rev Biochem.1987;22:1–38. 4. Porta M, Sassi M, Cavallazzi M, et al. Tourette’s syndrome and role of tetrabenazine. Review and personal experience. Clin Drug Invest. 2008;28: 443–459. 5. Jankovic J, Orman J. Tetrabenazine therapy of dystonia, chorea, tics and other dyskinesia. Neurology. 1988;38:391–394 6. Chen JJ, Ondo WG, Dashtipour K, Swope DM. Tetrabenazine for the treatment of hyperkinetic movement disorders: a review of the literature Clin Ther. 2012 Jul;34(7):1487-504 Article By-Dr Nischal Maskey MD Pediatrics Resident PICU IOM, TUTH
