Skin Tumors: Cause, Features and Morphology
Almost everyone develop skin tumors during his/her lifetime, but since most of them are benign they may not need treatment but are removed for cosmetic purposes. Since most of the skin neoplasms are visible, they are easily detectable.
Classification:
a. Benign Epithelial Tumors:
- Seborrheic Keratoses
- Acanthosis Nigricans
- Fibroepithelial Polyp
- Epithelial Cyst
- Adnexal Tumors
- Keratoacanthoma
b. Premalignant and Malignant Epidermal Tumors:
- Actinic Keratosis
- Squamous cell carcinoma
- Basal cell carcinoma
- Merkel cell carcinoma
c. Tumors of Dermis:
- Benign Fibrous histiocytoma (Dermatofibroma)
- Dermatofibrosarcoma Protuberans
- Xanthomas
- Dermal Vascular Tumors
d. Tumors of Melanocytes:
e. Tumors of cellular immigrants:
- Langerhans Cell Histiocytosis
- Cutaneous T-Cell lymphoma
- Mastocytosis
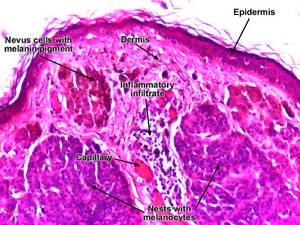
Melanocytic Nevus (Mole)
- Any congenital or acquired neoplasm of melanocytes
Morphology:
Types:
- Junctional nevi:
- nevus cells grow in aggregates (nests) along the dermoepidermal junction
- nuclei: uniform, rounded, inconspicious nuclei and show little or no mitotic activity
- Compound nevi:
- most junctional nevi grow into underlying dermis as nests or cords of cells to form compund nevi
- Intradermal nevi:
- In older lesions, epidermal nests may be lost entirely to form pure intradermal nevi

Less mature nevus cells: superiorly placed, larger, melanocytic and grow in nests
More mature nevus cells: placed deeper, smaller, no or little pigment and grow in cords
Dysplastic Nevi
- larger than most acquired nevi (>5mm)
- usually show variegation (variability in pigmentation) and irregular borders
- tendency to occur on non-sun exposed as well as on sun-exposed body surfaces
- may give rise to malignant melanoma
Morphology:
- Lentiginous hyperplasia: single nevus cells begin to replace normal basal cell layer along the dermoepidermal junction
- Cytologic atypia consisting of irregular, often angulated nuclear contours
- Sparse lymphocytic infiltrate
- Melanin pigment incontinence: phagocytosis of melanin pigment by macrophages
Malignant Melanoma
Sites: Skin, oral and ano-genital mucosal surfaces, oesophagus, meninges, eye
Predisposing factors:
Sunlight (light pigmented people at higher risk)
Pre-existing nevus (eg. dysplastic nevus)
Hereditary factors
Exposure to carcinogens
Clinical features:
Change in color, size, or shape in a pigmented lesion with irregularity in border
Itching or pain in pre-existing mole
Moles can also be a problem sometimes, but did you know that they can be removed? I visited a wonderful clinic that does mole removal in Manchester recently and they did an amazing job, so that is definitely worth considering if you have a problem with a mole.
Morphology:
Melanoma cells : larger than nevus cells, large nuclei with eosinophilic red nucleoli
i. Radial growth (Early Melanoma):
- horizintal growth within epidermal and superficial dermal layers
- no capacity to metastasize
- types: lentigo maligna, superficial spreading and acral/mucosal lentiginous
- melanoma cells proliferate as poorly formed nests or as individual cells at all levels of the epidermis
ii. Vertical growth (Advanced Melanoma):
- growth of melanoma downward into deeper dermal layers as an expansive mass lacking cellular maturation with time
- metastatic potential present
- melanoma cells grows as expansile, balloon like nodules in dermis
Squamous cell carcinoma
- malignant tumor of epidermal keratinocytes
- higher incidence in men than in women
Cause: mutation in p53 gene
Predisposing factors:
- Sunlight
- Industrial carcinogens
- Chronic ulcers and draining osteomyelitis
- Old burn scars
- Ionizing radiation
- Tobacco and betel nut chewing
Sites: Sun exposed sites usually in older people
Morphology:
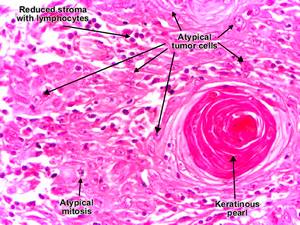
a. In-situ carcinoma:
- not invaded through BM (Basal Membrane) of dermoepidermal junction
- atypical nuclei at all levels of epidermis
- sharply defined, red, scaling plaques
b. Invasive lesions:
- when cells break through BM
- nodular, hyperkeratosis, ulceration
- variable differentiation:
- polygonal squamous cells with zones of keratinization
- highly anaplastic rounded cells with foci of necrosis
- abortive with dyskeratosis
Basal cell carcinoma
- slow growing tumors that rarely metastasize
- most common tumor arising on sun exposed sites
- pearly papules often containing prominent, dilated subepidermal blood vessels (telangiectasias)
- may contain melanin and appear similar to melanocytic nevi or melanomas
- rodent ulcers
Morphology:
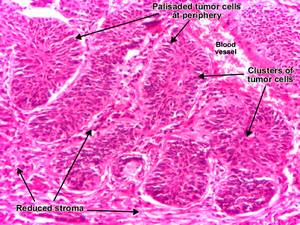
Resembles normal basal cell layer of epidermis
2 patterns of growth:
i. Multifocal: growing from epidermis to skin surface
ii. Nodular lesions: growing downward deeply into the dermis as cords and islands of variably basophilic cells with hyperchromatic nuclei; often surrounded by many fibroblasts and lymphocytes
Palisading arrangement of cells peripheral to tumor cells
Stroma shrinks away from epithelial tumor nests creating clefts or separation artifacts
Notes Adapted from Robbins Basis of Pathology