HIV with a Special Focus on Screening, Diagnosis and Monitoring
HIV: Basics of the disease
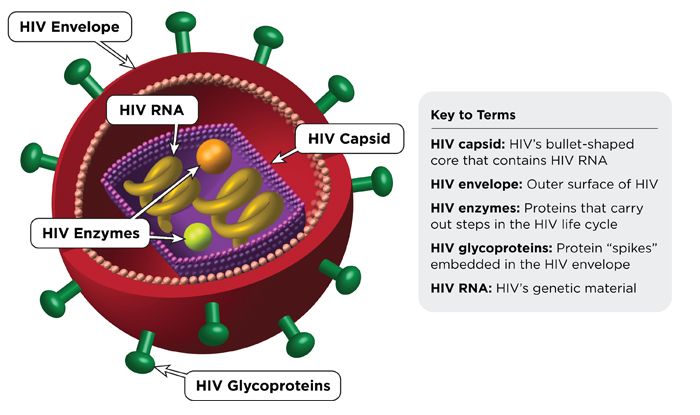
HIV or Human Immunodeficiency virus is a pathogen of the retrovirus family responsible for causing the HIV infection which subsequently develops into the highly dreaded and fatal disease called AIDS or Acquired Immunodeficiency Syndrome. HIV infection can be acquired upon contact with the body fluids of an infected individual. Unprotected sexual intercourse with an infected person, infection from infected pregnant mother to fetus and infection that spreads through the use of infected needles and syringes; comprise the various commonly known modalities of transmission of HIV infection.
HIV attacks the CD4 or T cells in the body. These cells constitute a key element in the body’s defence mechanism towards diseases, opportunistic and secondary infections in particular. As the copies of HIV in the bloodstream of the infected person keep increasing, one can expect a progressive decline in the CD4count. This in other words implies that with a growing viral load, the immune system of the body and the person’s ability to guard against infections and several other diseases; is progressively depleted. Opportunistic infections like tuberculosis and certain cancers like Kaposi’s sarcoma are common among HIV positive patients.
What’s the difference between HIV and AIDS?
HIV is a virus and causes an initial infection which begins to attack the immune system. HIV infection over a period of time may progress into AIDS; a full blown disease which increases the patient’s vulnerability to several disorders. AIDS stands for acquired immunodeficiency syndrome. AIDS is the final stage of HIV infection, and not everyone who has HIV advances to this stage.
AIDS is the stage of infection that occurs when your immune system is badly damaged and you become vulnerable to opportunistic infections. When the number of your CD4 cells falls below 200 cells per cubic millimeter of blood (200 cells/mm3), you are considered to have progressed to AIDS. (Normal CD4 counts are between 500 and 1,600 cells/mm3.) You can also be diagnosed with AIDS if you develop one or more opportunistic infections, regardless of your CD4 count.
Without treatment, people who are diagnosed with AIDS typically survive about 3 years. In case of a dangerous opportunistic illness, life expectancy without treatment falls to about 1 year.
From HIV to AIDS: Slow Progressors versus Fast progressors
The progression from HIV to AIDS takes place at a different pace for different patients. A small percentage of HIV-infected individuals rapidly progress to AIDS if they fail to take the medication within four years after primary HIV-infection and are termed Rapid Progressors. Indeed, some individuals have been known to progress to AIDS and death within a year after primary infection.
Another subset of individuals who are persistently infected with HIV-1, but show no signs of disease progression for over 12 years and remain asymptomatic is classified as Long Term Non-Progressors. In these individuals, it seems that HIV-infection has been halted with regard to disease progression over an extended period of time.
HIV/AIDS: Epidemiology
Globally, as per a fact sheet released by the UNAIDS in 2014:
People living with HIV
In 2013, there were 35 million [33.2 million–37.2 million] people living with HIV.
Since the start of the epidemic, around 78 million [71 million–87 million] people have become infected with HIV and 39 million [35 million–43 million] people have died of
AIDS-related illnesses
New HIV infections
New HIV infections have fallen by 38% since 2001.
Worldwide, 2.1 million [1 .9 million–2.4 million] people became newly infected with
HIV in 2013, down from 3.4 million [3.3 million–3.6 million] in 2001
AIDS-related deaths
AIDS-related deaths have fallen by 35% since the peak in 2005.
In 201 3, 1.5 million [1.4 million–1.7 million] people died from AIDS-related causes worldwide compared to 2.4 million [2.2 million–2.6 million] in 2005
India has the third largest HIV epidemic in the world. In 2013, HIV prevalence in India was an estimated 0.3%. This figure is small compared to most other middle-income countries but because of India’s huge population (1.2 billion) this equates to 2.1 million people living with HIV. In the same year, an estimated 130,000 people died from AIDS-related illnesses.1 Overall, India’s HIV epidemic is slowing down, with a 19% decline in new HIV infections (130,000 in 2013), and a 38% decline in AIDS-related deaths between 2005 and 2013. Despite, this 51% of deaths in Asia are in India.
The significance of screening for HIV among high risk groups and suspected individuals
Worldwide, it has been known and established that certain sections of society have a higher prevalence of HIV/AIDS. These include commercial sex workers, prostitutes, the lesbian/gay/transgender (LGBT) community, intravenous drug abusers, prisoners, truck drivers and migratory workers. Besides these vulnerable groups, the risk of HIV infection is higher among people who have unprotected sex with multiple sexual partners. It is in such high risk subgroups of society that HIV screening assumes tremendous significance. It helps early detection of HIV infection which can make treatment more effective and improve the overall outcome, quality of life and life span of an infected individual.
Tests to screen for HIV infection
HIV antibody testing is used to screen and diagnose HIV infections. Most people develop detectable HIV antibodies within 6 to 12 weeks of infection (this can take up to 6 months in rare cases). Rapid card tests are most commonly used for HIV antibodies detection; different immunoassay techniques mainly Enzyme Linked Immunoassay (ELISA) or automated immunoassays are also been used for detection of HIV antibodies Regardless of the type of screening test used, a positive result requires follow up with a second test to establish a diagnosis of HIV. Current testing protocols usually involve an initial screening by two different rapid card tests. If positive, the HIV antibody immunoassay test is repeated by more specific immunoassays. Recently, a combination of antibody and antigen test also known as 4th generation tests are used for early diagnosis. They are recommended four weeks after exposure as compared to antibody-only tests that are recommended after six weeks. 4th generation tests detect over 95% of infections at four weeks after exposure. If you are looking for a free hiv test, you may want to visit the nearest clinic in your area for more info.
Tests for monitoring the infection
Once a patient is diagnosed, following two key tests are used to monitor the infection:
CD4 count – CD4 lymphocyte cells (also called T-cells or T-helper cells) are the primary targets of HIV. The CD4 count and the CD4 percentage mark the degree of immunocompromise. It is used to stage the patient’s disease, determine the risk of opportunistic illnesses, assess prognosis, and guide decisions about the urgency of starting antiretroviral therapy (ART). The CD4 percentage is the percentage of the lymphocyte population that is CD4+; it is measured directly by flow cytometry. The mean normal value is approximately 500-1,300 cells/µL Illness, vaccination, diurnal variation, laboratory error, and some medications can result in transient CD4 cell count changes, whereas the CD4 percentage remains more stable. Because CD4 counts may vary, treatment decisions generally should not be made on the basis of a single CD4 value. A change between two test results is considered significant if it is a 30% change in absolute CD4 count or 3% point change in CD4 percentage. In an untreated HIV infection, the CD4 count declines by approximately 50-80 cells/µL per year.
HIV-1 Viral Load – HIV-1 viral load measurement indicates the number of copies of HIV-1 RNA per milliliter of plasma. Although HIV ultimately resides within cells, the plasma measurement is an accurate reflection of the burden of infection and the magnitude of viral replication. It is used to assess the risk of disease progression and can help guide initiation of therapy. It is critical in monitoring virologic response to ART. After initial infection with HIV, the viral load quickly peaks to very high levels, usually >100,000 copies/mL. Generally, 3-6 months after primary infection, the viral load declines and then remaining in a steady state. Among patients who are not on ARV medications, a small number maintain a low or even undetectable viral load, but the vast majority of those patients have relatively high HIV RNA levels. Higher plasma viral loads are associated with more rapid declines in CD4 cells and with increased risk of progression to symptomatic disease and AIDS; they also are associated with higher risk of HIV transmission. In untreated persons, the CD4 cell count is more useful in determining the urgency of ART initiation, but once a patient has started ART, the viral load is used to monitor the response to therapy. A key goal of ART is to achieve a viral load that is below the level of quantitation (e.g., <40 copies/mL). Because CD4 and clinical responses may lag behind changes in viral load, viral load testing is essential for detecting virologic failure in a timely manner. With an effective ARV regimen, a decline of at least 10-fold (1 logarithm) is expected within the first month, and suppression to undetectable levels should be achieved within 3-6 months after initiation of therapy. The HIV viral load should be checked at least twice at baseline, before the patient starts an ART regimen. Follow-up viral load measurement should be performed at regular intervals usually over 3-6 months intervals. Viral loads, like CD4 counts, are affected by laboratory variation, assay fluctuations, and patient variables such as acute illness and recent vaccinations. Variations of <0.5 log10 copies/mL (threefold) usually are not clinically significant. Recent illnesses or vaccinations can transiently increase viral load. If a patient has had a recent illness or vaccination, the viral load measurement should be deferred for 4 weeks, if possible. Different methodologies (Real Time PCR, NASBA, TMA, Automated Real Time PCR etc) are available for HIV Viral Load monitoring, due to inherent technical difference between these methodologies, ideally monitoring is done by using same technology and preferably same laboratory. Among available technologies US FDA approved automated Real Time PCR (Roche and Abbott) are most popular.
HIV Drug Resistance – As HIV multiplies in the body, the virus sometimes mutates (changes form) and produces variations of it. Variations of HIV that develop while a person is taking HIV medicines can lead to drug-resistant strains of HIV. Drug resistance can cause HIV treatment to fail. A person can initially be infected with drug-resistant HIV or develop drug-resistant HIV after starting HIV medicines. Genotypic testing is recommended as the preferred resistance testing to guide therapy in antiretroviral (ARV)-naive patients. Standard genotypic drug-resistance testing in ARV-naive persons involves testing for mutations in the reverse transcriptase (RT) and protease (PR) genes. Most genotypic assays involve sequencing of the RT and PR genes to detect mutations that are known to confer drug resistance. Interpretation of test results requires knowledge of the mutations selected by different antiretroviral (ARV) drugs and of the potential for cross resistance to other drugs conferred by certain mutations. Generally genotype resistance tests are recommended when first diagnosed and before most treatment changes. Usually they can detect mutations responsible for 24+ different antiretrovirals from 6 classess.
HIV vaccine: A hope for many
The development of an effective HIV vaccine that could either prevent HIV infection from occurring or would have a therapeutic effect on patients already infected, has been the quest of the medical and research fraternity globally, for several decades. Several large clinical studies have been conducted in the past and several are underway, to come up with a sustainable HIV vaccine. Unfortunately, research in this filed hasn’t borne any substantial success. This is largely due to the characteristic of HIV which allows it to mutate its form very rapidly. Millions of patients across the globe as well as the medical community are waiting hopefully for a potential vaccine to be offered as a robust solution to the HIV predicament. One should be careful of STDs and HIV. Fortunately, there are now HIV Prevention programs that can help people who are at risk of getting infected.
Featured Author

President-Research & Innovation,
Mentor-Molecular Pathology and
Clinical Research Services
SRL R&D, Mumbai