Genome Wide Association Studies & Personalized Medicine
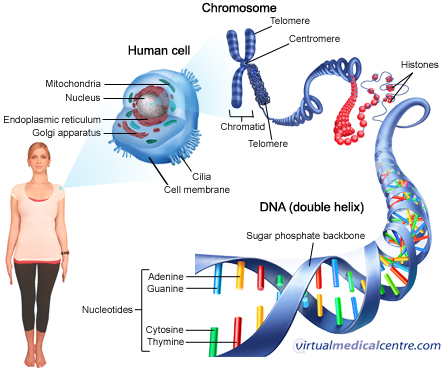
The study of drug metabolism is associated with genome wide association studies in which different gene clusters are studied for single nucleotide polymorphisms (SNP). The warfarin case clearly indicates that different individuals would show different responses because of SNPs.
From the drug development and research point of view, biomarkers are very important for the progress of personalized medicine. SNPs are also a type of biomarker that have very diverse applications, ranging from diagnosis, prognosis to the selection of targeted therapies.
Polymorphisms and bio-markers
From the clinical side of view, SNP is aiding pharmacodynamics and treatment monitoring. Genome wide association studies have enabled researchers to identify biomarkers and categorize them based upon the diseases and the appropriate function.
For instance, the Urinary Protein Biomarker Database is dedicated to biomarkers that are detected in the urine and can be used for both therapeutic and diagnostic measures. The PSA test for prostate cancer is a good application of the database.
The application of genome wide association studies have vastly attributed to biomarker and personalized medicine development for cancer. The cancer driver and drug development database can be accessed by researchers and drug developers to pursue various avenues in the treatment of cancer.
Doctors, as well researchers involved in drug development, may also utilize professional drug testing to acquire pharmacogenetics information about patients via PGT. This type of testing brings into consideration the SNPs within a population, allowing the user to monitor the efficiency of the therapy being developed more thoroughly.
The importance of genetic variation
The aspect of genome wide association studies (GWAS) is related to personalized medicine. The genetic variation within individuals is responsible for both drug susceptibility and the treatment response. So by scanning the genomes of individuals, genetic variants that lead to diseases can be pointed out.
In the same way, pharmcogenetic markers that would aid in treatment are also a product of associative studies. This has streamlined the scope of personalized medicine beyond that of the optimal drug response. Now it is possible to predict when to withhold treatment, provide preventive intervention and also proceed for targeted treatment options.
Personalized medicine has also helped researchers in identifying the the broad application of anti-inflammatory therapies. For instance, a recent study has highlighted that factors such as anti TNF and anti-IL-6 are effective against Crohn’s disease when normal therapy fails.
With the ever expanding scope of GWAS, researchers are developing prognostic and predictive biomarkers. These are pretty much going to redefine the treatment paradigm and also lead to advancement in personalized medicine. Prognostic biomarkers are associated with the prediction of the progress of the disease while the predictive biomarker gives an insight on the response to a treatment.
Personalized medicine is an important area of research, where academia and commercial industries are both trying to achieve market level success.
Article by Jennifer Smith