Celiac Disease: An Overview
Celiac disease (also known as celiac sprue or gluten intolerance) is an autoimmune disorder. Celiac disease occurs when the body’s immune system triggers a response to consumption of gluten, in genetically predisposed individual. The immune response damages the small intestine, which in turn interferes with the absorption of nutrients from food.
Gluten is the protein found in wheat which is responsible for the immune response in a person with celiac disease, but often other proteins such as hordeins in barley, and secalins in rye, can also result in an immune attack in susceptible individuals.
Statistics about celiac disease
Celiac disease (CD) affects approximately 1% of the population worldwide. In India celiac disease is more prevalent in the North Indian population, where wheat is primarily grown and forms the staple diet 1. An ICMR Task Force initiated study has indicated the prevalence of CD and potential CD as 8.53/1000 and 3.70/1000 in Haryana, 4.66/1000 and 3.92/1000 in Assam, and 0.11/1000 and 1.22/1000 in Tamil Nadu respectively 2.
The prevalence has been doubling every 20 years and several theories have been proposed to explain this alarming rise; changes in the dietary habits of people, usage of new strain of wheat which is more antigenic, as well as highly improved diagnostic tests1
It is also being said that though the diagnostics tests have improved and are highly sensitive to detect celiac disease, but the number of people getting diagnosed (3-5%), may just be the tip of the iceberg3. This may be due to the fact that the disease presentation is different in different age groups, and the classical presentation of malabsorbtion and chronic diarrhoea may present in only a fraction of the cases.
Signs and Symptoms
Only 35% of individuals receiving a new diagnosis of celiac disease present with diarrhea, abdominal pain, and bloating. The other signs and symptoms are:
| Signs and Symptoms | Associated Disorders |
| Short Stature , Delayed puberty, amenorrhea | Osteoporosis -due to a deficiency of vitamin D and calcium |
| Chronic fatigue | Bleeding disorders- due to altered vitamin K absorption |
| Depression, anxiety | Anemia- deficiency of either folate or iron due to alter ed nutrient absorption |
| Tooth enamel defects | Lactose intolerance-inability of intestinal villi to secrete lactase |
| Oral canker sores | Nervous system disorders, tingling hands or feet, seizures |
| Skin Rash (dermatitis herpetiformis) | |
| Arthritis, joint or bone pain |
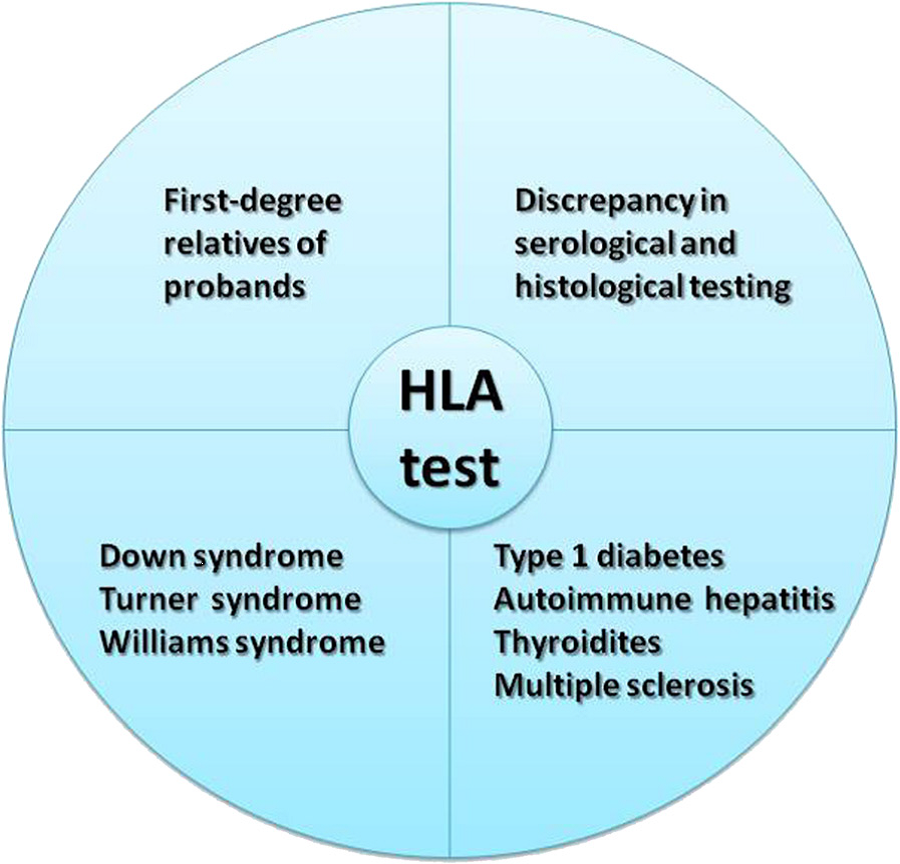
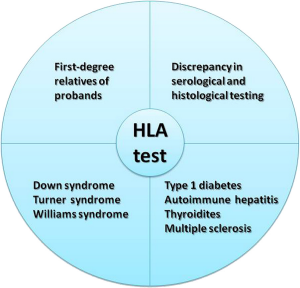
Some patients may also be asymptomatic. These patients may only receive a diagnosis when they undergo screening due to them being in a higher-risk group. The high risk include those who have a relative with celiac disease, those who have other autoimmune disorders (thyroiditis and multiple sclerosis), or those who have type 1 diabetes. First-degree relatives of patients as well as individuals with specific genetic syndromes (Down, Turner, Williams) are also at an increased risk.
Left undiagnosed and untreated celiac disease is a serious disease with a high mortality rate.
Diagnosis
Early diagnosis of celiac disease is critical and may prevent complications later in life.
The diagnostic assays for CD are
- Serological Assays- determine the level of antibodies present as a response to gluten
- Endoscopic Biopsy –the gold standard for confirming the diagnosis
- Genetic Tests – determine the presence of the predisposing HLA gene
Serological Assays, include anti tTG- IgA (tissue transglutaminase), anti-endomysial and gliadin (deamidated) antibody IgA. These antibodies are highly specific for CD.
Deviating a little into the pathogenesis of CD will help us understand why the serological assays are so specific. Gluten peptides pass through the epithelial barrier of the small intestine into the lamina propria, where they undergo enzymatic modificationby tissue transglutaminase. This process is called deamidation and leads to an increased immunogenicity of glutenpeptides. In the lamina propria, the gluten peptides are ‘recognized’ as foreign antigens and presented by particular HLAmolecules on antigen-presenting cells (APCs). This triggers a cascade of innate and adaptive immune responses and leads to the production of antibodies against gliadin – i.e. anti-endomysium (EMA) and anti-tissue transglutaminase antibodies- as well as to the production of pro-inflammatory cytokines.
Because of the high specificity of these assays often the need for small bowel biopsies is obviated, which is particularly advantageous in children. The IgA anti-tTG provides the best specificity. However, about 6 per cent of healthy individuals are partially IgA deficient, so a false negative result may be obtained if only the IgA antibody test is done. Recent diagnostic kits often provide both IgA and IgGantibody levels both against tTG and DGP (deamidatedgliadin protein), increasing the ability to make the diagnosis.
Endoscopic Biopsy – In CD the inflammatory response results in the destruction ofthe intestinal epithelium and mucosa, and lymphocytic infiltration in the proximal part of the small bowel. This tissue remodelling eventually causes flattening of the intestinal mucosa, villous atrophy and crypt hyperplasia.
Deep duodenal biopsies, obtained at upper gastrointestinal endoscopy, are often used to confirm the diagnosis by demonstrating infiltration of the epithelium by lymphocytes and the presence of villous atrophy. Biopsy should be performed before gluten-intake has been stopped4.
Genetic Tests, determine thepresence of the predisposing HLA (human leukocyte antigen) alleles HLADQ2 and DQ8 allele. More than 97%of celiac patients carry either HLA-DQ2 or HLA-DQ8 or both.
The HLA- DQ alleles are basically heterodimers composed of alpha and beta chains, in case of HLA-DQ2, it is (DQA *501/DQB *201) and or HLA-DQ8 (DQ *301/DQ *302). These two serotypes have been reported in 13%-30% of different populations in India3.
HLA typing is routinely requested to provide additional support in dubious CD cases, i.e. uncertain or discrepant serology and/or biopsy, and in at-risk categories. HLA is a useful test in screening first degree relatives due to the higher prevalence of CD among relatives of celiac patients5
In a meta-analysis of 6 Western studies, the sensitivity of HLA-DQ2 and HLA-DQ8 typing for detection of CD was 98% [95% confidence interval (CI) 97-99] and specificity 45% (95% CI 41-48)6. If HLA-DQ2 or HLA-DQ8 was not detectable by typing, the negative likelihood ratio for CD was 0.05 (0.03-0.09). Due to its great sensitivity and low negative likelihood ratio, HLA-DQ2/DQ8 typing is very useful in ruling out CD. A negative HLA DQ2/DQ8 test makes it highly unlikely that the individual will develop the disease.
Recommendation:
Guidelines from the European Society for Paediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN) for thediagnosis and treatment of celiac disease indicate “HLA testing should be performed in patients with an uncertain diagnosis of CD, forexample, in patients with negative CD-specific antibodies and mild infiltrative changes in proximal small intestinal biopsy specimens. If CD is considered in children in whom there is a strong clinical suspicion of CD, high specific CD antibodies are present, and smallbowel biopsies are not going to be performed, then the working
group recommends performing HLA-DQ2 and HLA-DQ8 typing to add strength to the diagnosis. Alsoduodenal biopsy can be omitted in cases with elevated serum anti-TG2 antibodies (>10x upper limit of normal), positive EMA and at-risk HLA”7.
Treatment
A life-long gluten-free diet is the only available and effective therapy, which leads to normalization of histological and serological parameters and to complete remission of all clinical signs. Typically symptoms can improve within one month for 95% of patients who follow a gluten-free diet, healing of the intestinal villi can take 3-6 months in children. In adults, it can take years.
Adherence to gluten free diet is imperative and ingestion of even small amounts of gluten may result in symptoms and damage
To eat is human, to digest, divine. –Mark Twain
| A combination of genetics and exposure to gluten are the two necessary predisposing factors for the development of celiac disease. |
References
- Ramakrishna BS. Celiac disease: can we avert the impending epidemic in India?The Indian Journal of Medical Research. 2011;133(1):5-8.
- Ramakrishna BS et al . Prevalence of Adult Celiac Disease in India: Regional Variations and Associations. Am J Gastroenterol. 2016 .doi: 10.1038/ajg.2015.398.
- ICMR Guideline on Diagnosis and Management of Celiac Disease in India 2016
- Rubio-Tapia et al. ACG Clinical Guidelines: Diagnosis and Management of Celiac DiseaseAm J Gastroenterol 2013; 108:656–676.
- Megiorni and Pizzuti. HLA-DQA1 and HLA-DQB1 in Celiac disease predisposition: practical implications of the HLA molecular typing. Journal of Biomedical Science 2012, 19:88.
- Díaz-Redondo et al. The potential usefulness of human leukocyte antigen typing for celiac disease screening: A systematic review and meta-analysis. Rev Esp Enferm Dig. 2015;107:423-9.
- European Society for Pediatric Gastroenterology, Hepatology, and Nutrition Guidelines for the Diagnosis of Coeliac Disease.JPGN 2012;54: 136–160
Authors


R & D, SRL Limited
Prime Square, Plot No.1, S. V. Road
Goregaon (West), Mumbai 400062, India