Thalidomide induced Teratogenecity: Phocomelia
Thalidomide taken during Pregnancy, in 1960s caused a horror outbreak of a birth defects of limbs called Phocomelia
Labeled as pregnancy category X, the teratogenicity of thalidomide is most severe. Lipid soluble and readily crosses the placenta, so it should never be taken by pregnant women or those who could become pregnant.
Thalidomide was released in 1956 as a mild sedative used to combat nausea in pregnant women. Later in 1961 it was withdrawn from the market once it was discovered thalidomide was a human teratogen. Approximately 5,000-7,000 malformed infants were born to women who ingested thalidomide during pregnancy.
Thalidomide was released in 1956, in West Germany. As a sedative or hypnotic, thalidomide also claimed to cure “anxiety, insomnia, gastritis, and tension.” Later it was used against nausea and alleviate morning sickness in pregnant women. Thalidomide became a popular drug in Germany around 60s, and could be purchased without a prescription. Shortly, in Germany, between 5,000 – 7,000 infants were born with the qualities of phocomelia ( seal limbs).
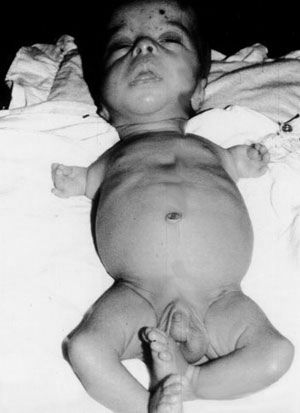
Among the children born with phocomelia, only about 40% of them survived.Reseach showed the direct cause was linked to thalidomide which significantly increased the incidence of phocomelia. The statistic was given that “50 percent of the mothers with deformed children had taken thalidomide during the first trimester of pregnancy.”
Shortly, throughout Europe, Australia, and the United States, 10,000 cases were reported of infants with phocomelia; only 50% of the 10,000 survived.Thalidomide became effectively linked to death or severe disabilities among babies. Those subjected to thalidomide while in the womb experienced limb deficiencies in a way that the long limbs either weren’t developed or presented themselves as stumps. Other effects included: deformed eyes, hearts, alimentary, and urinary tracts, along with blindness and deafness.
Even one dose of a 50 mg capsule can cause severe birth defects. The teratogenic risk is highest during the critical period, which is days 20-40 of gestation or days 35-50 after the last menstrual period
Teratogenic effects of thalidomide
- fetal limb growth retardation (arms, legs, hands, feet)
- ingrown genitalia
- absence of lung
- partial/total loss of hearing or sight
- malformed digestive tract, heart, kidney
- stillborn infant

How such drugs were allowed to be clinically used without proper trials.
Human experiments should be prohibited and such drug companies should be punished.