The golden era of genomics and its application in human diseases
Background:
Background:
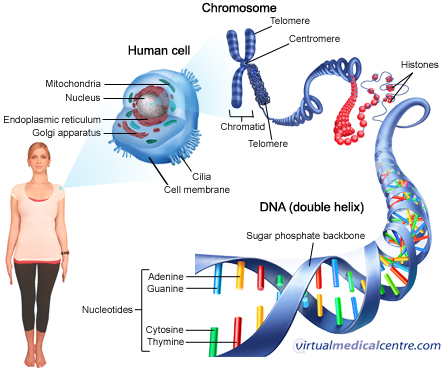
Genomics is an interdisciplinary field of science focusing on the structure, function, evolution, mapping, and editing of genomes. The milestone discovery of the human genome sequence in early 2000 was the first step in understanding how the instructions coded in DNA lead to a functioning human being. We have slowly moved ahead and taken the next step to derive meaningful knowledge from the DNA sequence. Now at this juncture of genomic research, we can confidently say that virtually every human ailment has some basis in our genes. Because of advancement in genomic knowledge, researchers and doctors have more powerful tools to study the role that multiple genetic factors acting together and with the environment play in much more complex diseases such as cancer, diabetes, and cardiovascular disease across the globe. This has enabled scientific community and medical practitioner to design and develop improved and early diagnostics, better treatment regimens, and evidence based strategies for demonstrating increased clinical efficacy with reduced side effects. Let us understand some critical application of genomics in various aspects of human diseases.
Diagnosis of rare monogenic disorders
As of now, there are over 7000 individually rare monogenic diseases which contribute to a significantly high rate of morbidity, mortality and health care expense. The greatest irony of the disease is that even after extensive investigations to identify the specific cause of a rare disease, the underlying aetiology remains unidentified for a very large number of cases. Although application of various genomic tools such as chromosomal microarrays have increased the diagnostic yield, but the diagnosis still remains unclear in patients with a presumed monogenic aetiology. Recent advanced cutting-edge genomic technologies such as Next generation sequencing (NGS) has empowered us to sequence the entire genome by whole genome sequencing approach as a throw away cost. Many genomic based laboratories in India as well as globally have adapted this technology for identifying the possible genetic cause the rare disease more efficiently. Global reports suggests that application of genome-wide sequencing have significantly improved the diagnostic rate to the tune of 25 to 30% with whole exome sequencing in a range of patients suggestive of monogenic disease.
Prenatal testing
Genetic diseases are often devastating and may cause significant disability and even death in childhood. Prenatal testing consists of prenatal screening and prenatal diagnosis, both of which allows detection of problems associated with the pregnancy and consequently make decisions about whether to continue with the pregnancy. Current method of prenatal screening consists of blood-based biochemical testing or ultrasound measurements or both, combined with maternal age. Furthermore, an invasive diagnostic test (e.g. amniocentesis) is done in those women who are at high risk for fetal aneuploidy. These methodologies have their own limitations with respect to low sensitivity and specificity (55-85% accurate) and invasive nature of testing which increases chances of miscarriage (4-5% cases). Interestingly, genomics-based non-invasive prenatal testing (NIPT) using NGS method has bought a revolution in prenatal screening testing. It can easily identify fetal aneuploidy as well as microscopic deletions across the genome with an accuracy of 99% from the blood of pregnant women. This testing can be done across a wide range of gestational age starting from as low as 10th week till 20th weeks of pregnancy. This has significantly bought down the number of invasive amniocentesis due ti its high sensitivity and specificity. This test is now currently available in advanced genomics labs in India and abroad as well.
Cancer predisposition and treatment:
The study of cancer genomes has revealed abnormalities in genes that drive the development and growth of many types of cancer. Genomic based studies have enabled us to understand the role of genetic abnormality in cancer predisposition as well as cancer treatment. Certain genetic variants that are often inherited from a parent increase the risk of developing a particular type of cancer. These genetic changes contribute to the development of a disease but do not directly cause it. Some people with a predisposing genetic variation will never get the disease while others will, even within the same family. Example of genes which predisposes an individual to cancer includes mutations in the BRCA1 or BRCA2 genes which greatly increase a person’s risk of developing breast cancer and ovarian cancer. Similarly, mutation in APC gene increases the risk for developing colon cancer, SMAD4 mutation for prostate cancer so on and so forth. These genomic variations can be easily detected simultaneously by the help of NGS technology and thereby helping for timely intervention who are at risk of developing cancer. Another aspect of genomic based testing is targeted therapy, which is nothing but a newer type of cancer treatment that uses drugs or other substances to more precisely identify and attack cancer cells. It is now possible to identify different actionable mutations across different cancer type which will ultimately help the clinician to decide the best suited therapy with maximum benefit and lesser side effects. Few examples are EGFR gene mutation in lung cancer, KRAS gene in colon cancer and BRAF mutation in melanomas. Infact with the help of NGS based testing; it is possible to identify the clinically relevant mutation in more than 50 genes along with the most appropriate drugs available for the patients. Some of the most commonly available panels for predisposition and treatment are hereditary cancer panels and tumor hot spot panels.
Infectious diseases:
Infectious diseases are one of the leading causes of death across different parts of the globe. It is imperative to have accurate and quick diagnostic methods for optimal clinical management of the infected patients. However, even till today, the gold standard for the diagnosis of infectious diseases has long been the culture which includes isolation, identification and antibiotic-susceptibility testing of the causative microorganism from patient sample. One of the major limitations of this diagnostic scheme is the time taken (minimum of 24 hours). This trend is slowly replaced by genomic based testing (PCR, RT-PCR, Realtime PCR etc) which is less labor intensive, more sensitive and gives results within hours. In addition, advanced molecular testing such as genome sequence analysis has enabled the development of various clinical-microbiology tools for pathogen detection, identification or genotyping at genus, species, or strain level. In addition it also allows rapid detection of genes associated with antibiotic resistance or virulence which aids in optimal therapeutic decision making and better patient management. Furthermore, with high throughput NGS sequencers, it is possible to sequence the bacterial whole-genome (WGS) to understand the sequence-based discrimination across different strains. The usefulness of such investigations has already been demonstrated, wherein WGS was used to compare 86 human M. tuberculosis isolates from a German outbreak and was found to be superior over other genotyping methods for tracing and investigating micro-epidemics. Furthermore, identification of multidrug resistance pattern seems to be another promising area in genomic based testing.
Future of genomics based application:
Clearly, genomics based approach has lots of promises, and looks like “the genomic era” is now a reality. In contemplating a vision for the future of genomics research, it is appropriate to consider the remarkable path that has brought us here. We have witnessed a tremendous improvement in patient management by applying our knowledge of genomics and new technology used in treatment will continue to do in near future, an example of new technology would be In-home therapeutic Virtual Reality.
Author Information